Management
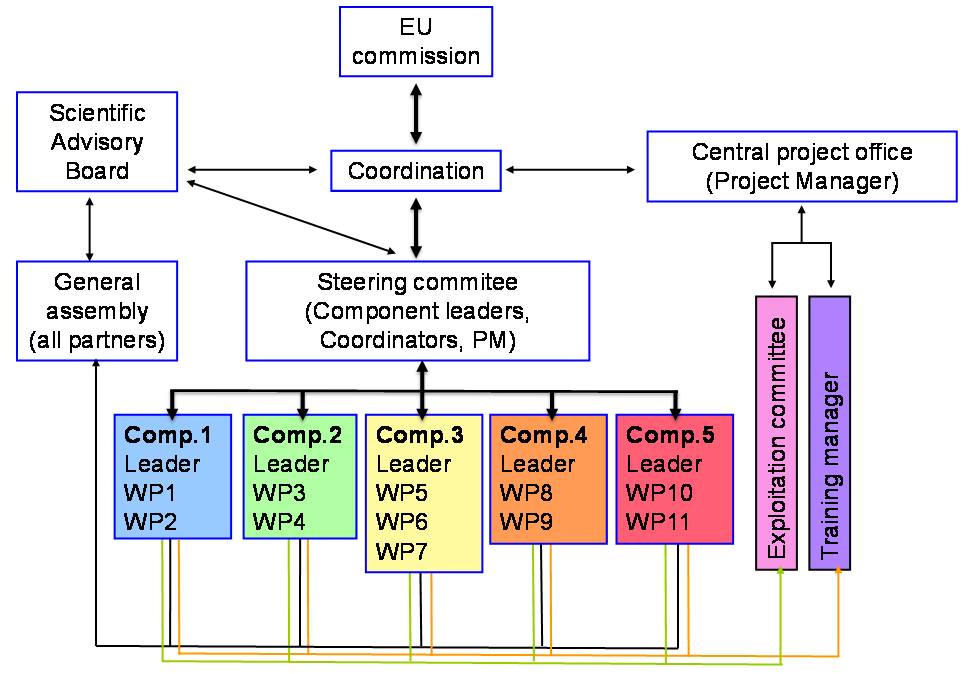
Schematic of SYSCILIA Organisation
Project Coordinator and a Co-coordinator
The Coordinator functions as the official representative of the project and as intermediary between the consortium and the Commission as well as supervisor of the Central Project Office. The Co-coordinator will participate in all coordination activities and will assume general coordination should the Coordinator be committed to other activities.
Central Project Office
The Central Project Office is based at the Radboud University Nijmegen Medical Centre. It will support the Coordinator in the day-to-day operational management of the project and is responsible for day-to-day project administration.
The Central Project Office consists of:- Coordinator: Ronald Roepman
- Project Manager: Simone Dusseljee
- Project Administrator: Marion Bussemakers
- Financial Officer: Dennis Vissers
- Legal Officer: Maarten van Langen
All information related to the project shall be transmitted to the Commission through the Central Project Office.
General Assembly (GA)
Each partner organization is represented by one person in the GA, preferentially the responsible PI or his/her delegate. The General Assembly will meet to decide on vital issues of the consortium. It will meet on demand of the Steering Committee or the Coordinator. The coordinators function as chair(s) of the GA. Participating personnel from each laboratory can attend the annual meeting of the GA in addition to the principal investigators.
Work Package Leaders
Each work package is under the responsibility of a single partner, the work package leader. The WP Leader organizes the appropriate contacts between the partners concerned and are in charge of producing the deliverables.
Component Leaders
The five Component Leaders will act as scientific coordinators of the individual components in collaboration with the WP leaders. The component leaders will ensure timely coordination of inter WP activities especially with respect to ensuring an integrated conductance of wet and dry lab activities. They will coordinate the flow of data between the partners and the relevant WP to ensure exchange of data and integration between different component and WPs.
Steering Committee (SC)
The SC will specifically address strategic and contractual deci-sions concerning the project. It will be headed by an elected chairman and comprise one component leader of each component, one member of the Exploitation committee, and the Project Manager to effect strategic decisions concerning all scientific and financial aspects of the project including the detailed work program and scientific controlling. The SC will make use of external scientific advisors (Advisory Board) (3-4 scientists) who are not partnering the Consortium.
External Advisory Board (EAB)
The EAB will critically monitor the conductance and performance of the project. The EAB will be invited to all Annual Network Meetings, they may participate in component meetings to give independent advice to the project. In addition to critically monitoring the scientific conductance of the project, the EAB will monitor the composition of the offered career opportunities and training activities and make sure that the recruited young researchers will acquire skills which are essential for career development in both the academic and industrial sector.
Members:

- Prof. Veronica van Heyningnen
- Medical and Developmental Genetics
- Medical Research Council, Human Genetics Unit
- Edinburgh, UK

- Prof. Dr. Alfred Wittinghofer
- Emeritus Group
- Max Planck Institute of Molecular Physiology
- Dortmund, Germany

- Prof. Martijn Breuning
- Clinical geneticist
- Leiden University Medical Center
- Leiden, The Netherlands

- Prof. Dr. Erich E. Wanker
- Proteomics and Molecular Mechanisms of Neurodegenerative Diseases
- Max Delbrueck Center for Molecular Medicine
- Berlin-Buch, Germany
Exploitation Committee (EC)
The Exploitation Committee will assess the possibility of translating the results obtained in the consortium. It will communicate with manufacturers, and will manage intellectual property rights (IPR).
Members:- Gordana Apic (relations to pharmaceutical industry)
- Ronald Roepman (patients organizations)
- Marius Ueffing (technology transfer)
- Heymut Omran (clinical medicine)
- Phil Beales (patient organizations)




